- Shanghai Zhongshen International Trade Co., Ltd. - Two decades of trade agency expertise.
- Service Hotline: 139 1787 2118
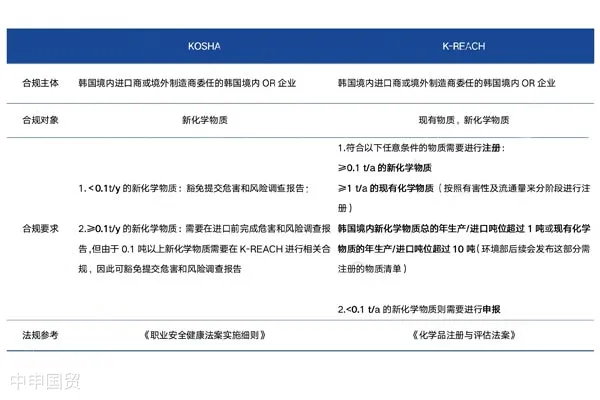
On July 19, 2023, the Ministry of Environment of South Korea announced that it would revise the Implementation Rules of the Chemicals Registration and Evaluation Act, mainly involving changes in the requirements for the transfer of chemical registration numbers in the supply chain.The revised regulations will come into effect on August 1, 2023.

According to the new regulatory requirements, after manufacturers/producers complete the substance registration/declaration and transfer substance registration - related information downstream, they need to submit relevant MSDS under KOSHA and transfer the hazard information in Document No. 26 of the K - REACH Regulations. The regulatory revision has updated the way registered substances/declared substances are reflected on the MSDS, but does not clearly specify the way of reflection, and the EUs approach can be referred to.
Changes in Regulatory Revision

Comparison of the Original Regulatory Texts:

This regulatory revision is considered to protect the trade secrets of enterprises, such as preventing the exposure of mixture composition information and avoiding the leakage of registration numbers/declaration numbers, etc., to avoid the leakage of enterprise information and loss of interests. Therefore, before importing into South Korea, enterprises should verify their products, confirm the substance information in the products and the product MSDS, and respond to South Korean regulatory systems in a compliant manner.
What are the compliant operations for substances declared or registered in South Korea?
Related Recommendations
? 2025. All Rights Reserved. 滬ICP備2023007705號-2  PSB Record: Shanghai No.31011502009912
PSB Record: Shanghai No.31011502009912